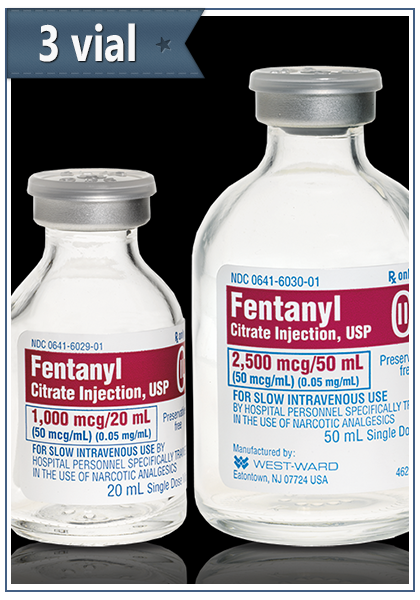
Fentanyl Citrate Injection 3 vials per package
$104.97 – $149.97Price range: $104.97 through $149.97
price per 1 package
Fentanyl Citrate Injection, USP is a sterile, nonpyrogenic solution of fentanyl citrate in water for injection. Fentanyl Citrate is a potent narcotic analgesic which is administered only by the intravenous or intramuscular routes of injection.
Minimum order quantity is: 1 package
Indications for Fentanyl Citrate Injection:
Fentanyl Citrate Injection 3 vials per package, premedication, induction, and maintenance; and in the immediate postoperative periods as the need arises. Analgesic supplement in general or regional anesthesia Concomitantly with neuroleptics (eg, droperidol) as an anesthetic premedication, for anesthesia induction, Fentanyl Citrate Injection 3 vials per package and as an adjunct in the maintenance of general and regional anesthesia. As an anesthetic in combination with oxygen in selected high-risk patients (eg, undergoing open heart surgery or complicated neurological or orthopedic procedures).
Adult:
Individualize. Premedication: 50–100 mcg (1–2 mL) by IM inj 30–60 mins before surgery. Adjunct to general anesthesia: see literature. Adjunct to regional anesthesia: 50–100 mcg (1–2 mL) by IM inj or slow IV (over 1–2 mins), when needed. Post-op: 50–100 mcg (1–2 mL) by IM inj; may repeat in 1–2 hrs as needed. General anesthetic: 50–100 mcg/kg (1–2 mL/kg) may be administered with oxygen and a muscle relaxant; in certain cases (eg, open heart surgery, complicated neurological or orthopedic procedures), doses up to 150 mcg/kg (3 mL/kg) may be needed. Elderly or debilitated: reduce the dose.
Children:
<2 years: not recommended. 2–12 years: induction and maintenance: 2–3 mg/kg.
Warnings/Precautions:
Should be administered only by persons specifically trained in IV anesthetics and respiratory management in an adequate facility. Have an opioid antagonist, resuscitative and intubation equipment, and oxygen readily available. Respiratory depression; monitor closely (esp. post-op). Comatose patients with head injuries or brain tumors Increased intracranial pressure. Hypertension. Pulmonary disease. Renal or hepatic impairment. Cardiac bradyarrhythmias. Drug abuse. Monitor vital signs routinely. Elderly. Debilitated. Premature neonates. Labor and delivery are not recommended. Pregnancy (Cat. C) Nursing mothers
Interactions:
Caution: within 14 days of MAOIs, Cardiovascular depression with nitrous oxide and high-dose fentanyl Concomitant droperidol may result in a decrease in pulmonary arterial pressure and hypotension. Additive effects with other CNS depressants (eg, barbiturates, tranquilizers [eg, diazepam], narcotics, general anesthetics]); reduce doses.
Pharmacological Class:
Opioid.
Adverse Reactions:
respiratory depression, apnea, skeletal muscle rigidity, bradycardia, hypertension, hypotension, dizziness, blurred vision, nausea, emesis, laryngospasm, and diaphoresis.
Related products
Painkillers
Painkillers
Painkillers
Painkillers
Painkillers
Painkillers
Painkillers
Painkillers